mulksgrp
Homer J. Simpson, The Simpsons 1995, S06E21.
Research in the mulksgrp
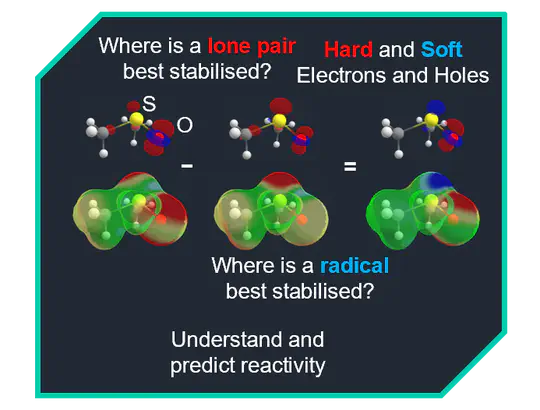
Renewable feedstocks for chemical synthesis are highly oxidised compared to crude oil. We aim to make these feedstocks more useful without energy-intensive processing through the development of reagents and catalysts based on highly oxidised organic molecules. We develop their uses as reagents and catalysts, for example, towards hydrodefluorination reactions, polymer degradations, and for capturing small molecules. We also develop computational models for cost-efficient and intuitive reactivity predictions in p-block elements.
We are a young independent research group founded at the Institute for Organic Chemistry (iOC) of the RWTH Aachen University in Germany in March 2022. Aachen is an atmospheric old German city in immediate vicinity of the Netherlands and to Belgium. The RWTH Aachen University is Germany’s largest university of technology and ranks 99 worldwide across all subjects and 66 in chemistry in the 2024 QS ranking.
We are always open to discuss science or working or studying with us. Get in touch!
Featured recent contributions

Funding
We greatly appreciate the support listed in the following!


